Summary of Scientists Uncover the Hidden Mechanism Behind FDA-Approved Alzheimer’s Drug:
A recent study by scientists from VIB and KU Leuven elucidates how the Alzheimer’s drug lecanemab (Leqembi) works by engaging microglial cells in the brain to clear toxic amyloid plaques. The researchers found that the antibody’s Fc fragment is essential for activating microglia, enabling them to effectively remove plaques associated with cognitive decline. This study, published in Nature Neuroscience, provides the first mechanistic insight into lecanemab’s efficacy and paves the way for safer and more effective Alzheimer’s therapies. The findings highlight the potential for future therapies to activate microglia without relying on antibodies, guiding the development of next-generation Alzheimer treatments.
*****
Key Takeaways
- Understanding Lecanemab: Lecanemab, an FDA-approved monoclonal antibody, targets amyloid plaques in Alzheimer’s and slows cognitive decline.
- Microglial Activation: A key discovery reveals that the Fc fragment of the antibody triggers the activation of microglia, crucial for clearing toxic amyloid plaques.
- Mechanistic Insights: The study provides a clear mechanistic understanding of how lecanemab functions, enhancing the development of future therapies.
- Research Significance: Findings pave the way for innovative Alzheimer’s treatments by understanding the specific microglial programs involved in plaque clearance.
- Collaborative Breakthrough: Conducted by the VIB-KU Leuven research team, the research underscores the importance of collaboration in scientific advancements.
Uncovering the Mechanisms of Lecanemab: A Potential Game Changer in Alzheimer’s Research
Alzheimer’s disease is a formidable foe, with over 55 million individuals affected globally. Characterized by progressive cognitive decline, it is closely linked to the accumulation of amyloid plaques in the brain. These plaques consist of misfolded proteins that disrupt neuronal function and development. The race to find effective treatments has been extensive, but the recent breakthrough regarding the FDA-approved drug, lecanemab, shines a hopeful light on this dark path.
The Role of Amyloid Plaques
Before diving into the specifics of lecanemab, it’s essential to understand the nature of amyloid plaques. These sticky aggregates, primarily composed of amyloid-beta proteins, accumulate in the spaces between neurons. They provoke inflammation and lead to neuronal cell death—an ominous scenario for anyone diagnosed with Alzheimer’s disease. The question then arises: How can we effectively mitigate this damaging buildup?
Lecanemab: A Step into the Future
Enter lecanemab, marketed as Leqembi. This monoclonal antibody has garnered attention for its ability to target and reduce amyloid plaques, thereby slowing the progression of cognitive decline in Alzheimer’s patients. What makes Lecanemab stand out from other treatments is its dual functionality: it not only binds to amyloid-beta but engages with the immune system to facilitate effective clearance of these toxic deposits.
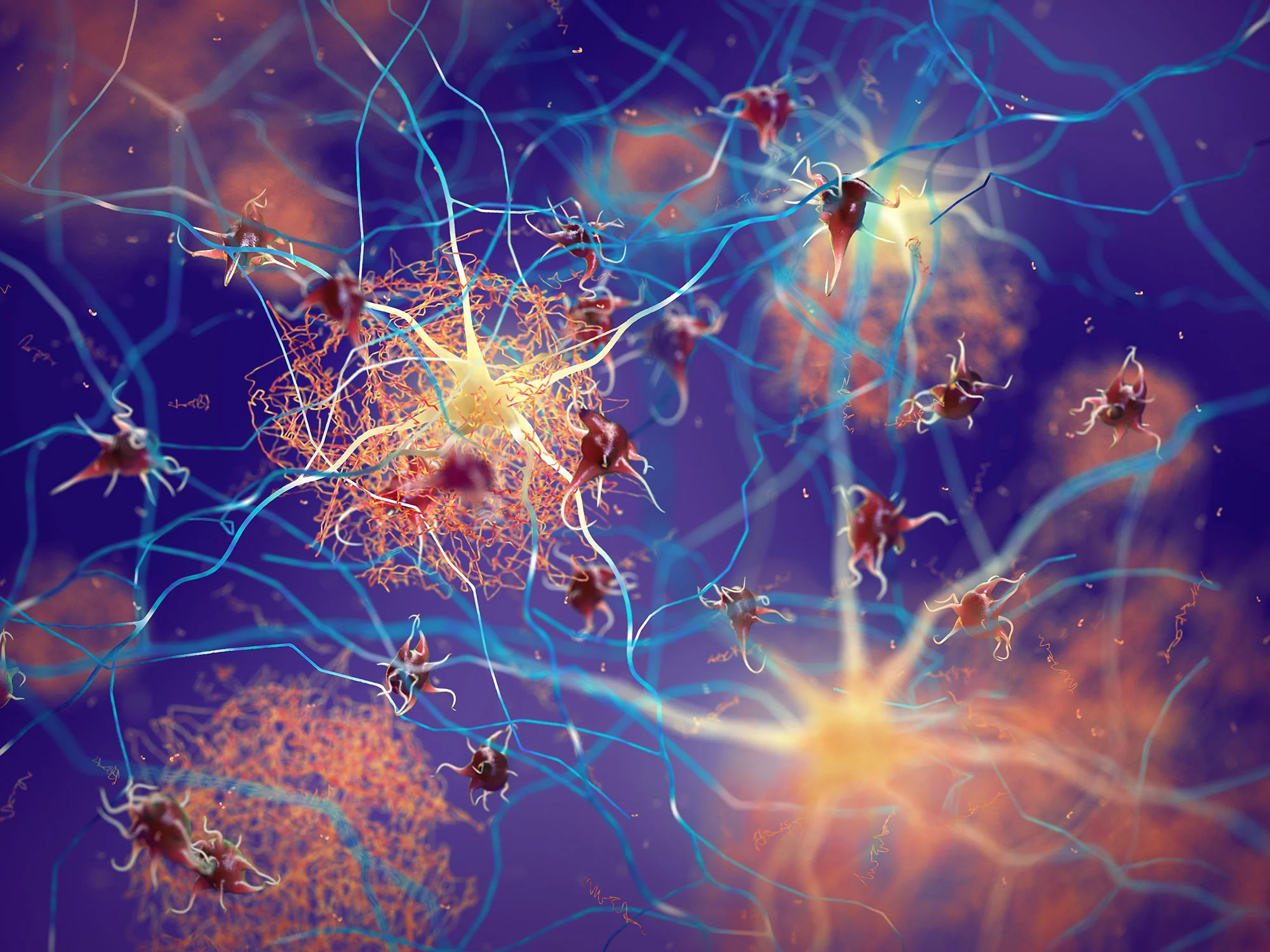
Microglia: The Brain’s Cleanup Crew
Microglia, the immune cells of the brain, play a critical role in monitoring the health of neural environments. Under normal circumstances, they gather around amyloid plaques. However, these tireless defenders often struggle to clear the accumulation effectively. The implication is clear: enhancing the functionality of microglia could provide a pathway to better outcomes in Alzheimer’s treatment.
The Game-Changing Discovery
Researchers from VIB and KU Leuven embarked on a mission to demystify how lecanemab operates. Their groundbreaking study unearthed that the drug’s effectiveness relies significantly on a particular part of the antibody known as the Fc fragment. This fragment acts like an anchor for microglia, enabling them to latch onto the amyloid plaques.
Dr. Giulia Albertini, one of the study’s co-first authors, emphasized how unique this finding is. “The therapy’s efficacy relies on the antibody’s Fc fragment, which activates microglia to effectively clear amyloid plaques,” she stated.
This statement not only captures the essence of the research but also highlights the transformative potential of understanding the mechanisms behind existing treatments. By engaging microglia through this Fc fragment, scientists can better harness the body’s natural cleanup processes—a significant leap forward in the realm of Alzheimer’s treatments.
Exploring the Science
To delve deeper, the researchers employed an innovative hybrid model: Alzheimer’s disease mice engrafted with human microglial cells. This meticulous setup allowed them to observe how lecanemab activates these human cells and facilitates amyloid plaque clearance. They discovered that the absence of the Fc fragment means no observable effect—an essential detail for future drug design.
“Using human microglia within a controlled experimental model was a major strength of our study,” remarked Magdalena Zielonka, another co-first author. This collaborative effort sheds light on the human-specific responses that could be instrumental in shaping future therapies.
Unraveling Cellular Processes
So, how exactly do microglia clear amyloid plaques? The scientists identified two core processes essential for this function: phagocytosis (the engulfing of debris) and lysosomal activity (degradation of unwanted materials). Notably, these processes kicked into gear only when the Fc fragment was present. This revelation sets the stage for further exploration into other potential therapies that could activate microglia without the need for antibodies—ultimately refining approaches to tackling Alzheimer’s.
The Bigger Picture: Future Implications
The implications of this research extend well beyond academic interest; they harbor the potential to revolutionize Alzheimer’s treatment. As we piece together how microglia contribute to plaque removal, it opens doors to therapies that might harness or enhance this function without the complexities involved in monoclonal antibodies.
Prof. Bart De Strooper wrapped up the conversation succinctly. “Understanding the importance of the Fc fragment helps guide the design of next-generation Alzheimer’s drugs.” In essence, the insights gleaned from this study not only illuminate the pathways that lead to effective therapies but also inspire a sense of hope in the ongoing battle against Alzheimer’s disease.
Motivation in the Face of Challenges
Breaking through scientific barriers can often seem daunting, much like facing a chronic illness. Yet this research embodies a compelling story of perseverance and collaboration. Each step taken by the research team underscores the importance of collective effort, innovative thinking, and the relentless pursuit of knowledge.
For those affected by Alzheimer’s, whether as patients, caregivers, or loved ones, these insights may offer not only hope but a reminder of the tireless work being conducted to improve lives. Sometimes, it is through the darkest struggles that the most significant breakthroughs arise.
Embracing the Future
As we forge ahead, the intersection of immunology and neuroscience offers tantalizing possibilities. The work done by VIB and KU Leuven stands as a beacon for future research, illuminating the path for refining therapies that could change lives.
In exploring the mechanisms behind existing medications, we can cultivate a richer understanding, fueling the next wave of creativity in drug development. Your journey—whether personal or professional—can likewise be seen through the lens of discovery. Each challenge you encounter might hold the key to a profound breakthrough; it’s about looking beyond the immediate obstacles to envision the possibilities that lie ahead.
Conclusions
The unearthing of the Fc fragment’s critical role in lecanemab’s function heralds a new era in Alzheimer’s research. With promising insights paving the way for enhanced therapies, it’s crucial to maintain a sense of curiosity and determination. After all, scientific endeavors reflect our broader experience of life itself—one of ongoing connection, exploration, and endless potential.
As we digest these remarkable findings, let us embrace a mindset centered around inquiry and resilience. Intelligence is born from asking the right questions; let this new understanding not only guide future research but inspire all of us to persist in our quests for knowledge and improvement, both personally and in our communities.
In closing, the revelation about lecanemab is not merely about a drug; it embodies the pursuit of hope, the power of science, and the unwavering spirit of human endeavor.