Summary of Scientists Uncover Hidden Blood Pattern in Long COVID:
Researchers have found a link between microclots and neutrophil extracellular traps (NETs) in the blood of individuals with Long COVID, potentially explaining persistent symptoms. Microclots are abnormal clusters of blood clotting proteins, while NETs are part of the immune response that can lead to inflammatory and clotting disorders when produced excessively.
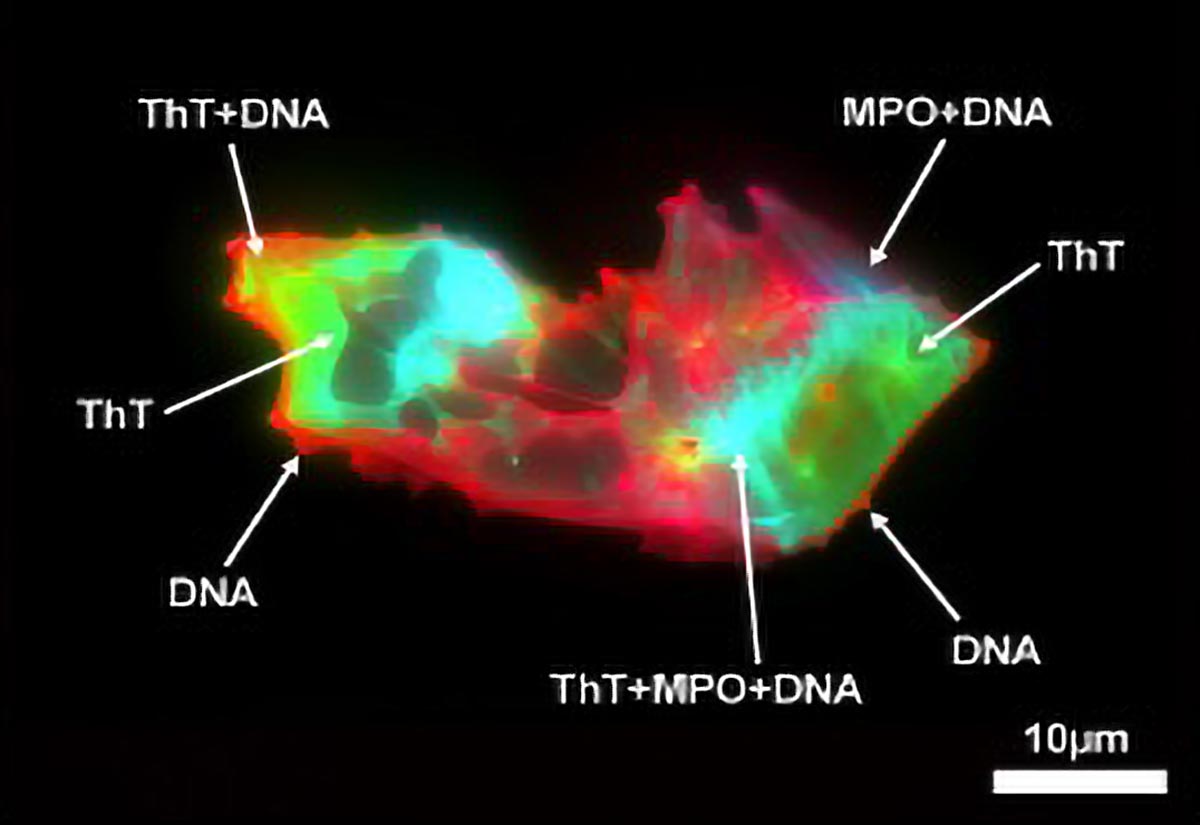
In their study, researchers observed significantly increased levels of both microclots and NETs in Long COVID patients compared to healthy individuals. They noted that these microclots were not only more numerous but also larger, suggesting a concerning interaction between microclots and NETs that may become pathogenic.
Advancements like machine learning were used to distinguish between Long COVID patients and healthy controls and identify potential biomarkers for treatment. These findings underscore the need for therapies that address abnormal clotting and inflammation, providing valuable insights for diagnosing and monitoring post-viral conditions.
*****
Key Points Summary
- Discovery of Microclots: Researchers have identified persistent microclots in the blood of Long COVID patients, which may explain ongoing symptoms.
- Neutrophil Extracellular Traps (NETs): These structures play a critical role in the immune response but can contribute to inflammation and clotting when overproduced.
- Research Collaboration: A joint study by multiple teams has revealed a strong link between microclots and NETs in Long COVID cases, suggesting they may affect health outcomes.
- Use of AI in Diagnostics: Advanced technology, including machine learning, enhances the accuracy of distinguishing Long COVID patients from healthy individuals, paving the way for personalized treatments.
- Implications for Treatment: This research opens avenues for targeted therapies to reduce harmful clotting and inflammation in Long COVID patients.
The Hidden Patterns in Long COVID: Microclots and NETs Unveiled
As we navigate the ever-evolving landscape of health, one of the most intriguing discoveries has surfaced in the realm of Long COVID—a condition that has baffled both patients and healthcare providers alike. Scientists have unearthed a hidden pattern involving microclots and neutrophil extracellular traps (NETs), components of our blood that could explain the lingering symptoms experienced by many. In this exploration, we’ll delve into the fascinating elements behind these findings, the vital roles these structures play, and the promising implications for future treatments.
What Are Microclots?
Imagine tiny clumps floating in your bloodstream, composed of proteins that normally serve to form protective clots after injuries. This concept lies at the heart of microclots. First identified by Professor Resia Pretorius in 2021, these abnormal aggregations have garnered significant attention for their potential association with various health complications stemming from COVID-19. Understanding microclots isn’t just a scientific curiosity; it’s a crucial piece of the puzzle concerning long-lasting health issues faced by COVID survivors.
The revelation that microclots exist and persist in Long COVID patients offers us a glimpse into pathways that might be obstructing recovery. Their dense structure forms a roadblock, making it difficult for blood to circulate effectively and thereby limiting oxygen flow to cells. This can lead to feelings of fatigue, brain fog, and other debilitating symptoms.
The Role of NETs in Our Immune System
Now, let’s pivot our focus to the remarkable role of NETs. These structures arise from a process called NETosis, where immune cells known as neutrophils unleash strands of DNA embedded with enzymes, designed to trap and neutralize harmful pathogens. While this mechanism helps protect us during infections, its overproduction can spiral into trouble, contributing to inflammation and clotting disorders.
Dr. Alain Thierry and his team have illustrated the consequences of excessive NET formation, linking them to various conditions like autoimmune diseases and cancer. Imagine a system designed for protection becoming overwhelmingly reactive—that’s precisely what happens when NETs become dysregulated. They can form a vicious cycle, fueling further complications rather than quelling the threat.
A Joint Exploration: Microclots Meet NETs
Recent collaborative efforts from formidable research teams led by Prof. Pretorius and Dr. Thierry have embarked on a quest to determine whether microclots and NETs are linked in Long COVID patients. Using advanced techniques like imaging flow cytometry and fluorescence microscopy, the researchers meticulously assessed blood samples, hoping to uncover whether these two structures interact.
What they found was striking: biomarker levels associated with both microclots and NETs were significantly elevated in Long COVID patients. Not only were there more microclots, but they were also larger than those found in healthy individuals. The structural association between microclots and NETs was notably stronger in the Long COVID cohort, which prompted a reevaluation of how these components might interact in ways that could exacerbate post-viral symptoms.
This finding isn’t just an isolated revelation; it’s a game changer that propels us deeper into the intricate biology of Long COVID. According to Dr. Thierry, this interplay could be pathogenic when dysregulated, reinforcing the notion that we must pursue effective strategies to manage both microclots and NETs.
Harnessing AI for Enhanced Diagnostics
Enter the world of artificial intelligence—an ally that has stepped into the fray of medical science with fervor. In this study, AI tools were employed to bolster diagnostic accuracy, allowing researchers to differentiate between Long COVID patients and healthy individuals. The algorithms sifted through extensive data, identifying the most informative biomarker combinations and guiding possible avenues for personalized treatment pathways.
This synergy between biotechnology and human inquiry adds a profound layer of assurance: we are not merely identifying symptoms; we are seeking solutions grounded in evidence-based insights. With each click of a machine-learning algorithm, we edge closer to understanding the complex interactions at play in Long COVID, offering a glimmer of hope to those entrenched in its struggles.
Implications for Treatment and Future Biomarkers
So, where do we go from here? By uncovering the mechanisms linking NETs and microclots, we not only gain insight into the biological processes underlying Long COVID but also ensure that we’re opening the door to therapeutic avenues. Imagine treatments specifically designed to reduce harmful clotting and curb inflammation, transforming the landscape of how post-viral conditions are understood and managed.
In addition to affording potential treatments, the study advances our capability to identify new biomarkers, pivotal for diagnosing and monitoring Long COVID and similar post-viral syndromes. The confluence of advanced imaging techniques and machine learning confers robustness, substantially enriching the ongoing discourse surrounding these conditions.
A Broader Perspective: The Road Ahead
The excitement surrounding these interdisciplinary findings is palpable. They embody the spirit of science’s relentless march toward discovery, where each breakthrough can translate into enhanced quality of life for those suffering from Long COVID. The integration of technology, biology, and clinical observation creates a modern tapestry woven through with hope—where every patient’s story can lead to broader understanding and new treatment paradigms.
But beyond the science lies a deeper significance. As we learn from these findings, resilience emerges as a theme. Each challenge faced—from small microclots to the overwhelming nature of Long COVID—mirrors the tenacity demonstrated in the human spirit. Just as scientists innovate solutions to combat health issues, individuals crafting their paths through adversity remind us of our innate capability to adapt, evolve, and heal.
Final Thoughts
As we reflect on the transformative nature of these discoveries surrounding Long COVID, it’s crucial to remember that science is not merely a collection of facts; it is an ongoing narrative enriched by inquiry, perseverance, and hope. In uncovering the intricate dance between microclots and NETs, researchers have paved the way for meaningful interventions.
Moving forward, let us foster a culture of curiosity and resilience, supporting one another in our respective journeys—whether through research, personal health, or emotional well-being. The story of Long COVID is still being written, and with every new chapter, we venture closer to understanding, healing, and revitalization.
Together, we stand on the precipice of scientific advancement that may irrevocably change the understanding of post-viral conditions, creating a more empowered future for all. The narrative continues, as do the possibilities—and with determination and insight, we can illuminate the path ahead.